How Beet Sugar is Made - Boiling
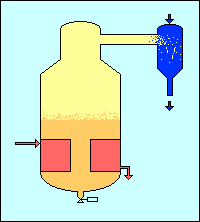
Physical chemistry assists with sugar purification during the crystallisation process because there is a natural tendency for the sugar crystals to form as pure sucrose, rejecting the non-sugars. Thus, when the sugar crystals are grown in the mother liquor they tend to be pure and the mother liquor becomes more impure. Most remaining non-sugar in the product is contained in the coating of mother liquor left on the crystals. This is washed off during the centrifugation.
In a beet factory nature is even kinder than in cane sugar processing because of the different nature of the colour chemicals in beet juice. In a cane factory one expects a 10:1 improvement in sugar colour from the syrup feed to the crystal product but in beet processing an improvement of as much as 100:1 is expected.
As in the cane processing, the mother liquor still contains valuable sugar so the crystallisation is repeated several times. However non-sugars inhibit the crystallisation. This is particularly true of other sugars such as glucose and fructose which are the breakdown products of sucrose. Each subsequent step therefore becomes more difficult until one reaches a point where it is no longer viable to continue. This is usually after three steps.



| 